Table of Contents
2. Who discovered californium?
3. Why is californium so expensive?
4. Where was californium discovered?
What is californium?
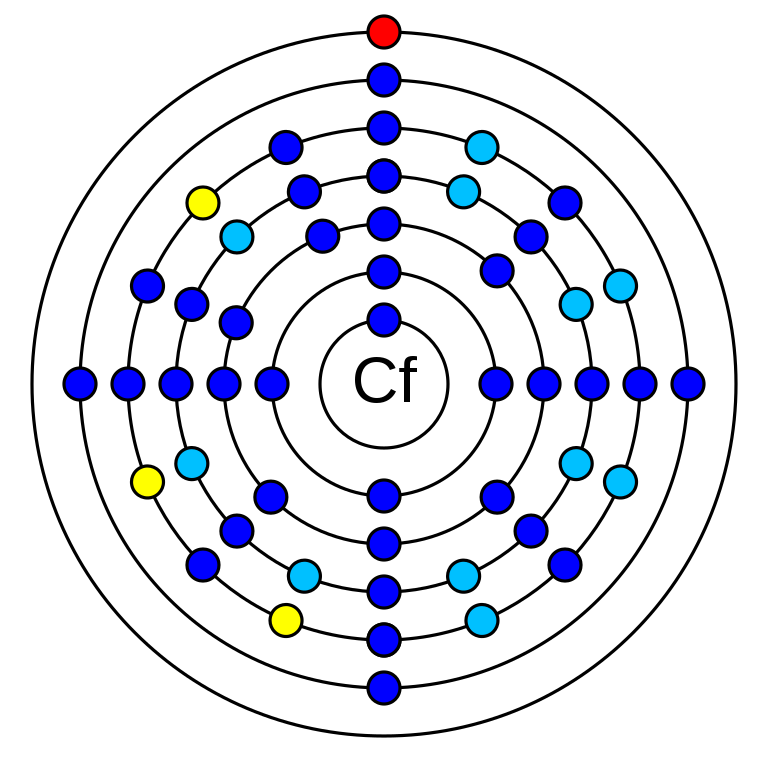
Californium is a chemical element that has an atomic number (Z) 98. Its mass number is 251 and it is represented by the symbol Cf. It is an actinide element in period 7 of the periodic table. It has 98 electrons and protons and 153 neutrons. Its electro negativity is 1.3 (Pauling scale). The electronic configuration of Californium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f14 5d10 6s2 6p6 5f10 6d0 7s2. Its oxidation states are +2, +3, +4, and +5. Californium is a synthetic element. There are some twenty radioisotopes of californium that have been synthesized.
Californium has a silvery appearance. At STP it is solid and has a density of about 15.1 g/cm3. It estimated melting point is 1173 K ?(900 °C, ?1652 °F) and the boiling point is 1743 K ?(1470 °C, ?2678 °F). It has two crystalline forms at standard atmospheric pressure: a double-hexagonal close-packed form dubbed alpha (α) and a face-centered cubic form designated beta (β).
Californium was discovered in Lawrence Berkeley National Laboratory in 1950 by a group of scientists (Stanley G. Thompson, Kenneth Street, Jr., Albert Ghiorso, and Glenn T. Seaborg). It was named after California, where it was discovered.
Although Californium is a synthetic element it can be found in radioactive dust and the fallout from atmospheric nuclear testing. It was once believed to be produced in supernovas.
It can be produced in nuclear reactors and particle accelerators. Californium-250 can be synthesized by bombarding berkelium-249 with neutrons producing berkelium-250 (via neutron capture). Berkelium-250 then turns into californium-250 by beta decays.
The extreme radioactivity making it very difficult to use for commercial and as well as laboratory uses. But it is a strong neutron emitter that can be useful as a neutron startup source for some nuclear reactors. Neutron moisture gauges use californium-252 to find water and petroleum layers in oil wells.
Who discovered californium?
Californium was discovered and synthesized by a group of scientists (Stanley G. Thompson, Kenneth Street, Jr., Albert Ghiorso, and Glenn T. Seaborg) in 1950. It was discovered in Lawrence Berkeley National Laboratory.
Why is californium so expensive?
Californium is expensive because it is hard to produce, a very small amount can be produced in years. It can be produced in a particle accelerator and a 60-inch cyclotron. As of 2003, Oak Ridge National Laboratory in the United States, and the Research Institute of Atomic Reactors in Dimitrovgrad, Russia produce 0.25 grams and 0.025 grams of californium-252 per year, respectively. This is how hard it is to produce that is why it is expensive. For commercial use, it is available through the U.S. Nuclear Regulatory Commission.
Where was californium discovered?
Californium was discovered at Lawrence Berkeley National Laboratory, California in 1950.