Table of Contents
2. How many electrons does curium have?
5. How many protons does curium have?
What is curium?
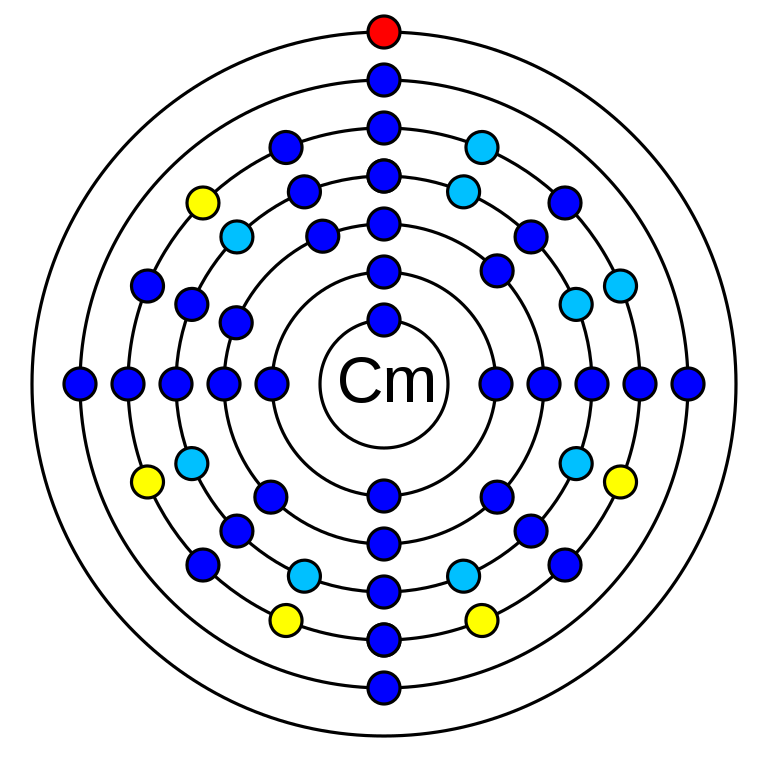
Curium is a chemical element that has the atomic number (Z) 96. Its mass number is 247 and it is represented by the symbol Cm. Curium is an element in the actinide series and period 7. It's an f-block element and has electro negativity 1.3 (Pauling scale). The oxidation states of Curium are +3, +4, +5, +6. The electronic configuration of Curium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f14 5d10 6s2 6p6 5f7 6d1 7s2. The ionization energy required for the removal of the first electron is 581 kJ/mol.
Keep in mind Curium is an artificial element. It has 19 known radioisotopes and 10 nuclear isomers.
At STP it has a density of 13.51 g/cm3. Its melting point is 1613 K ?(1340 °C, ?2444 °F) and its boiling point is 3383 K ?(3110 °C, ?5630 °F).
Curium was produced, isolated, and identified at the University of California, Berkeley in 1944. It was first intentionally synthesized by Glenn T. Seaborg, Ralph A. James, and Albert Ghiorso. It was named after Marie Sk?odowska-Curie (Marie Curie) and her husband Pierre Curie.
Curium isotope Cm-247 is a long-lived isotope that has a half-life of 15.6 million years. Maybe curium was present in Earth naturally in the early stages of its geological period. But later on, it decays to other elements. The most practical application of Curium is that of its isotope Cm-244. It is used as a source of α-particle in X-ray spectrometer.
How many electrons does curium have?
The Curium atom has 96 electrons.
What is curium used for?
For commercial uses, it is dangerous to use and expensive to produces. Its only practical use is in X-ray spectrometer as a source of α-particle.
Who discovered curium?
Curium was discovered by a group of scientists (Glenn T. Seaborg, Ralph A. James, and Albert Ghiorso) in 1944 at the University of California, Berkeley.
How many protons does curium have?
Curium has 96 protons in its nucleus.