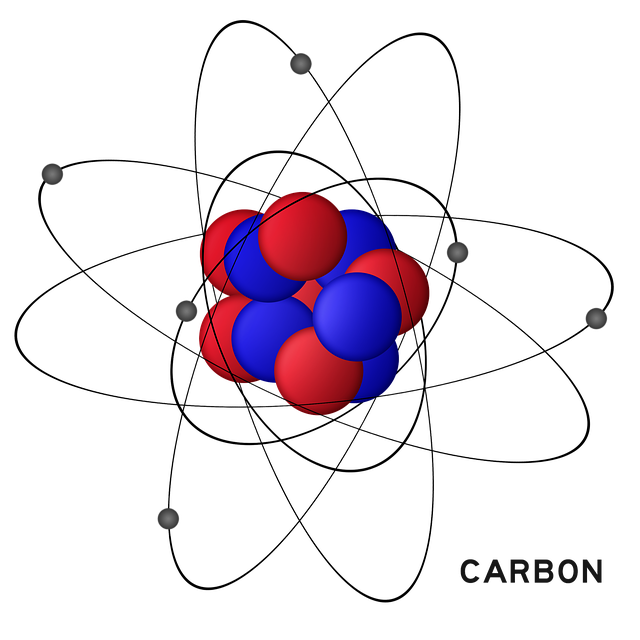
The electronic configuration of the Carbon atom is [He] 2s2 2p2. Carbon has six electrons. Two are in 1s orbital, two are in 2s orbital and the remaining two are in the 2p orbital. Carbon is a tetravalent atom meaning it has 4 electrons in its valence ready to deploy for covalent bonding with other atoms.
How many neutrons does carbon have?
Carbon has 6 neutrons, There are three naturally occurring isotopes of carbon. Two of them Carbon-12 and Carbon-13 are stable while carbon-14 is a radioisotope. C-12 has six neutrons, C-13 has seven while C-14 has eight neutrons. C-12 has an abundance of 98.9% while C-13 is just 1.1%.
How many protons does carbon have?
The atomic number or proton number of carbon is (symbol Z) 6, the number of protons in carbon is six. There are 6 electrons and 6 neutrons.